 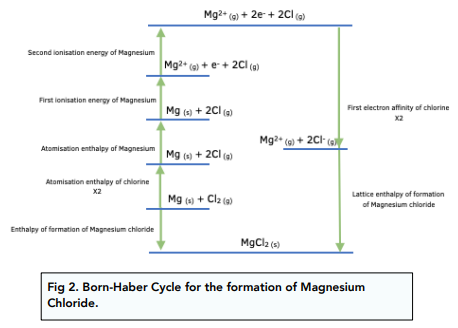
If the other energy terms of the cycle are known, they can be calculated from this cycle. Estimation of enthalpy of formation: One of the energy terms used in the Born-Haber cycle is the enthalpy of formation.The inter-electronic repulsion between the anion and extra electrons causes an issue in second electron affinity calculations. However, atoms can only be estimated to have a first electron affinity. Estimation of electron affinities: From theoretical lattice energy, the Born-Haber cycle can be utilized to calculate the electron affinities of individual atoms.Moreover, it determines the stability of the compounds. Stability of compounds: If different thermochemical data for a compound are substituted in the Born-Haber cycle, it will help to rationalize the existence of certain compounds and the non-existence of others.ΔH f = ΔH s + I (I 1 +I 2 +I 3) + 3/2 ΔH d + 3E + U (For MX 2 type salt) Application of Born-Haber Cycle Now, applying Hess’s law of constant heat of summation, the equation for enthalpy of formation for one mole of AlF 3(s) is given as: The formation of AlF 3(s) in two different ways is shown in the Born-Haber cycle: Hence lattice energy can be calculated by substituting the other energy terms in the Born-Haber cycle or in the above equation. The net enthalpy of formation and the first four of the five energy terms can be determined experimentally. – ΔH f = ΔH s + I + 1/2 ΔH d – E – U (For MX type salt) Therefore taking sign convention, negative for exothermic and positive for endothermic, the equation becomes: In the above equation, ΔH f, E, and U are exothermic terms, and the remaining three ΔH s, I and ΔH d are endothermic terms. ΔH f = ΔH s + I + 1/2 ΔH d + E + U (For MX type salt) Applying Hess’s rule, it is clear from the cycle that the enthalpy of formation (ΔH f) is equal to the algebraic sum of each of the five energy terms, or, in other words, the Born-Haber cycle equation for MX-type salt is Hess’s law of constant heat of summation states that the energy of the initial state (i.e., reactant) and the final state (i.e., product) in a chemical reaction determines the enthalpy change, but not the path traveled. The energy released in this process is called lattice energy(U). (e) Combination of Na +(g) and Cl –(g) to form NaCl(s): Gaseous Na + and Cl – combine to form crystalline NaCl(s). (i) Direct combination of Na(s) and Cl 2(g) The crystalline NaCl(s) can be formed by either of the following two different processes: The formation of NaCl(s) from two different paths interrelating different thermochemical quantities is shown in the cycle below: Let us take the formation of one mole of NaCl(s) (MX type salt) from Na(s) and Cl 2(g). How to calculate lattice energy from Born-Haber cycle? It was named after and developed by two German physical chemists Max Born and Fritz Haber who first used it in 1919.īorn-Haber cycle calculates the lattice energy by comparing the standard enthalpy of formation of the ionic compound (from the element) to the enthalpy required to make gaseous cations and anions from the element. Since the direct calculation of lattice energy is quite difficult as the required data is often not available, lattice energy is determined indirectly by the use of the Born-Haber cycle. It involves the formation of an ionic compound from the reaction of a metal (often group 1 and 2 elements) with a non-metal. The Born-Haber cycle is a simplified and cyclic method to correlate the lattice energy of ionic crystals with other thermodynamic data which is experimentally determinable.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
